 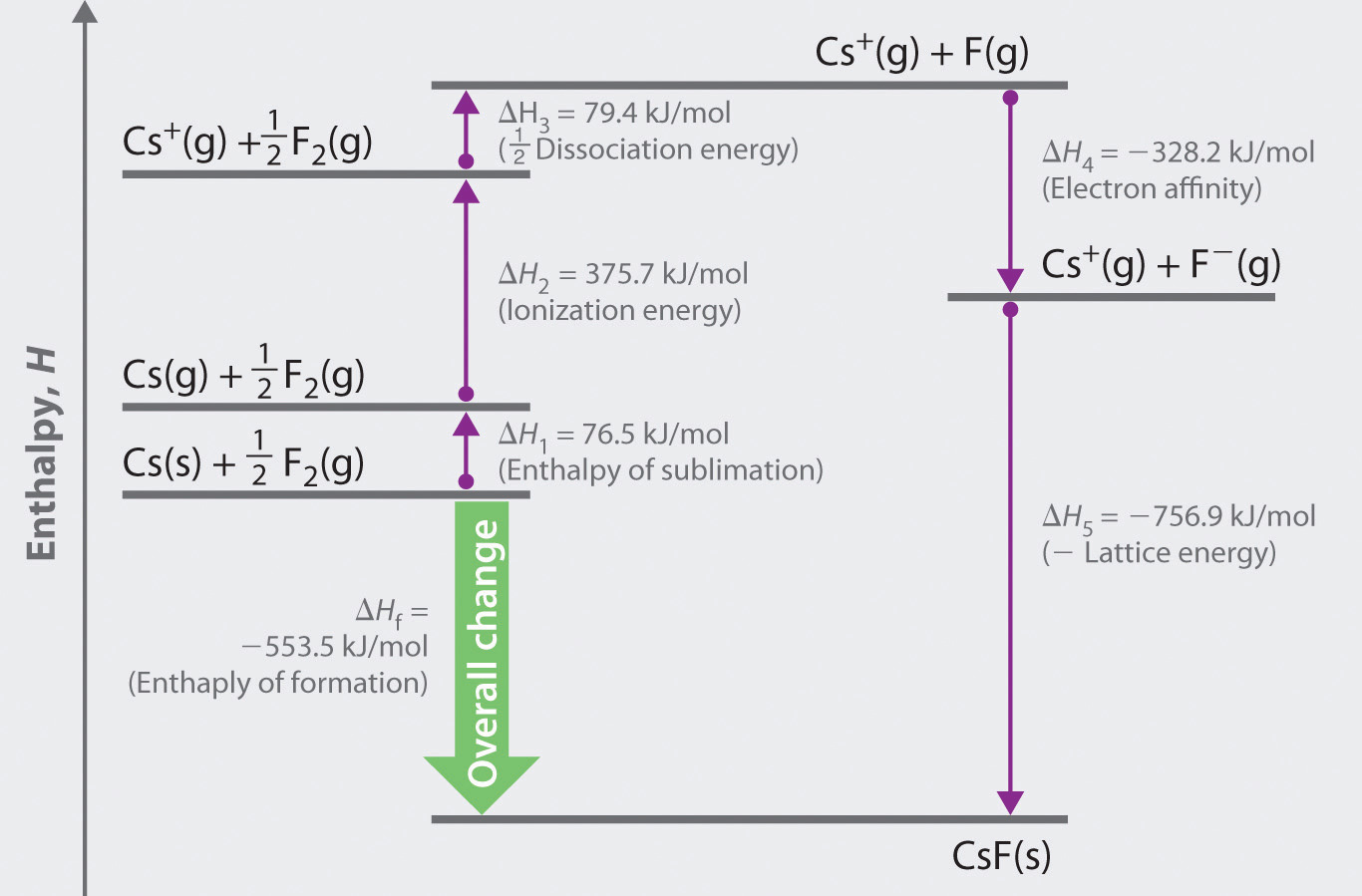
We begin with the elements in their most common states, Cs( s) and F 2( g). Lattice H denotes the molar lattice enthalpy. Where: LatticeU denotes the molar lattice energy. enthalpy changes needed to construct a Born-Haber cycle for sodium oxide. 
Give the appropriate sign for the energy change. The molar lattice energy of an ionic crystal can be expressed in terms of molar lattice enthalpy, pressure, and change in volume via the following equation: LatticeU LatticeH pVm. Calculating Lattice Energies The lattice energy of nearly any ionic solid can be calculated rather accurately using a modified form of Equation 4.1: U kQ1Q2 r0, where U > 0 U, which is always a positive number, represents the amount of energy required to dissociate 1 mol of an ionic solid into the gaseous ions. changes needed to calculate the lattice enthalpy of calcium oxide, CaO. In the table below, write an cquation for each reaction step in the process and give the associated energy change. \): The Born-Haber cycle shows the relative energies of each step involved in the formation of an ionic solid from the necessary elements in their reference states. Use the energies below to calculate the lattice energy of sodium oxide. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
